HAI, healthcare associated infections or hospital infections, pose a major challenge to public health systems, because they are a rather heterogeneous set of different conditions from a microbiological, physiological and epidemiological point of view that have a high impact on health costs and are indicators the quality of the service offered to hospitalized patients. The Elite device combines modern absorption media and highly effective filter materials; in fact, it uses several stages of filtration interconnected in falling with each other. The coordinated actions of Hepa filters, electric filter, germicidal action of antibacterial UV lamps, ozone action and catalyst filters guarantee the reduction of 99.9% of the PM and of the bacterial charges present in the open air, with a consequent decrease in the “infectious risk”. The Elite device was tested in a BCM room at the Lorenzo and Ariosto Seragnoli Institute of Hematology and Medical Oncology – S. Orsola Hospital in Bologna: it proved highly effective in breaking down very quickly (and keeping very low) the particulate pollution values influenced by the comings and goings of personnel, thus decreasing the infectious risk. In the test carried out in an intensive care room of the Policlinico di Piacenza, in very complicated environmental conditions, Elite determined in just 40 minutes of operation a reduction of 24-27% of the most significant particles, therefore an improvement in air quality .

Introduction
It is known that the microparticles (dust) contained in the air can pose a serious threat to the our health also in function of the fact that they represent the vehicle of transport in the environment for microorganisms (bacteria, viruses, fungi, spores etc ..).
Elite can therefore find a important use at the hospital level in all those situations in the which it is essential to bring and keep the microbial load as low as possible environmental; when possible, reset it. For example: BCM rooms, halls operations, intensive therapies, etc. The first test was carried out in period August – September 2018 in conjunction with renovation works upstairs. The Elite appliance was placed in one BCM room which housed a patient undergoing a transplant marrow.
The size of the room was 5m x 4m x 2.8m H, while the volume was 56m3. The flow rate of Elite is 1200 mc / h, in practice the device gives rise to 21.43 recycles / hour. After delivering the documentation relating to Use and Maintenance and instructing the nursing staff to operate the equipment, we positioned the Elite against a wall in the room, next to the hospital bed.
Materials and methods
For the first test we used:
Bio Aerosol 11-B
With the following characteristics:
Real time determination:
- Spores, Bacteria, Pollen, Fungi.
- Distribution particles in 31 classes.
- Count particles.
- PM 10, PM 2.5, PM 1, PTS.
- Partition particulates in: Inhalable, Thoracic, Alveolic.
Combined analysis of all particles measured on the slide, for fluorescence microscope analysis (not applied). Count mode from 0.25 µm to 32 µm
Mass mode from 0.1 to 100,000 µg / m³ We used a Personal PC, HP Compaq 8510w, for monitoring and instant saving of the acquired data.
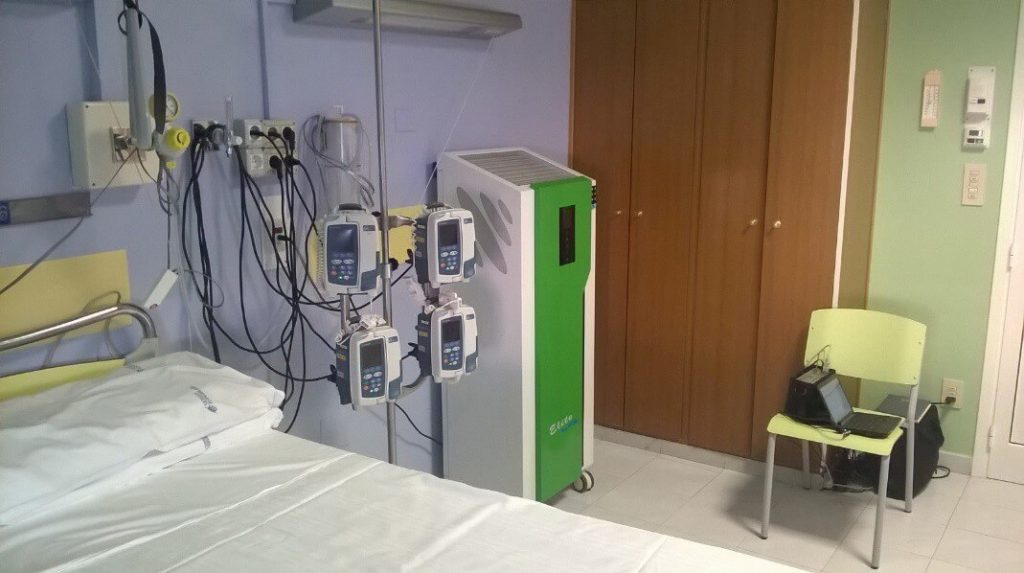
Positioning of the Elite device in the BMC room
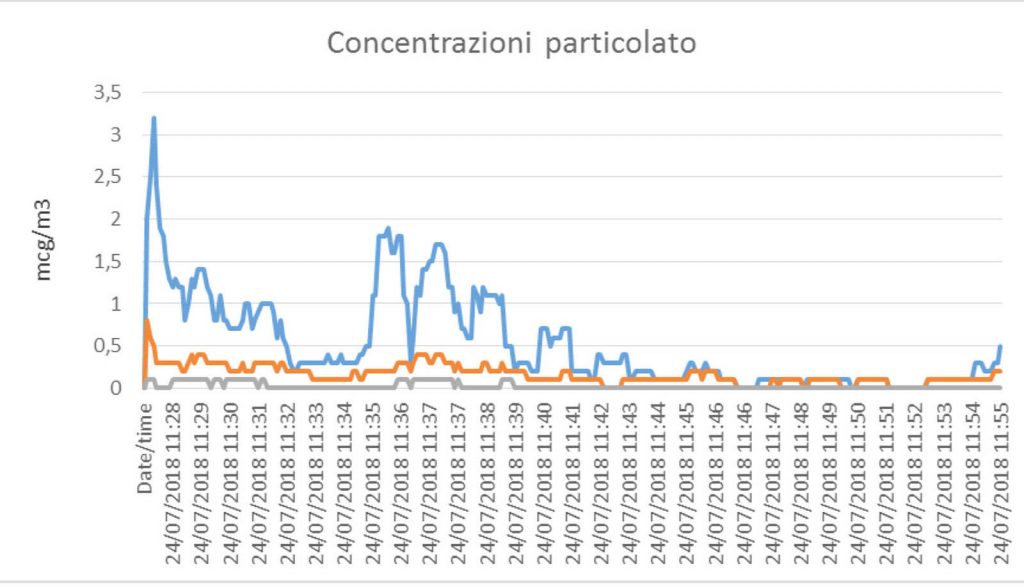
It highlights the rapid abatement of inert particles (and consequently microbial and mycetic load) after the rise due to the presence of medical and nursing staff
For the second test carried out at the Policlinico di Piacenza, the monitoring system consists of a Particle Counter Kanomax 3910 with a range of 50 LPM and 1 of CFM and sensitivity of 0.3 microns, placed on a support, provided by the compartment, and moved after each measure in another position to cover a representative area of the operating room / recovery room / environment under examination. The number of measurements is established by legislation, based on the type of technical standard indicated and based on the surface of the environment considered.
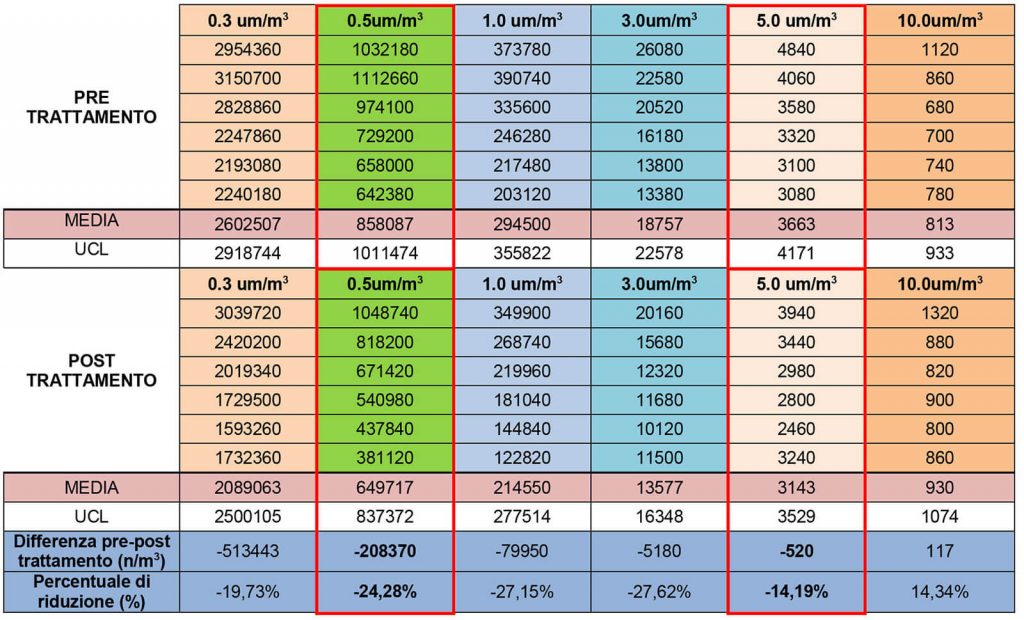
The elimination of the most significant inert particles (and consequently microbial and mycetic load) is highlighted after only 40 minutes of activity of the Elite device
Results
In the first test, the Elite continuously monitors PM 2.5 in real time. In order to have for this test data more precise and complete, the device was connected to an additional Bio Aerosol 11-B equipment which via program dedicated has allowed the reading of an important series of parameters. We have also requested and obtained a test carried out by a third party (Arpae) for the search for the total mycetic charge following the operation of Elite.
The duration of the second test, carried out by the Pollution Hospital Srl company, was 40 minutes. It was conducted to verify the abatement capacity of air-dispersed particulate obtained by a transportable system of recirculation and filtration of the ambient air. The survey was carried out in two phases: the first cycle of measurements was carried out prior to the use of the ELITE instrument: the second cycle of acquisitions (conducted in the same manner and with the same number of samples, in the same points of measurement) was performed after a first phase of air purification performed by the same filtration instrument. The investigation concerned the timely verification of the quantities of particles transported in the air. For the classification of air cleaning, the international standard EN ISO 14644-1 (classes 1 to 9) is used.
Conclusions
Elite has proven highly effective in quickly breaking down (and keeping very low) the values of inert particles influenced by the comings and goings of the nursing staff. The device therefore allows, by monitoring PM 2.5, a continuous and real-time control of the values of the particles and therefore also of bacteria, fungi and spores. Elite is small in size (cm.52x36x1.40H), easy to position and move and very simple to use.
Bibliography
- Azara A., Masia M.D., Maida I. et al. / Environmental pollution in operating rooms / View & Review, January 2000 p. 4-10
- Haley R.W., Hooton T.M., Culver D.H. et al. / Nosocomial infections in US hospitals, 1975-67. Estimated frequency by selected characteristics of patiens / Am. J. Med., 70 (1981); p. 947-958
- Indoor pollution microbiologico / I quaderni di Arpa – 2002
- Institute Superior for Occupational Prevention and Safety (ISPESL) / Guidelines for the definition of the safety and environmental hygiene standards of the departments operators / 1999
- Moro M.L. et al. / Risk factors for surgical wound infections in clean surgery: a multicenter study / 1995
- Pitzurra M., Pasquarella C., D’Alessandro D., Savino A. / Risk prevention in operating room / Universe Publishing Company – 1999
- Curls R., Lombardi R. / Protection from biological agents in the operating room: significance of the microbiological characteristics and type of interventions in some companies in the Marche region / Regional conference: Safety in companies Healthcare: Comparing Experiences – 2002
- K. Peters, R. Unger, A.M. Gatti, E. Monari, J. Kirkpatrick Effects of nano-scaled particles on endothelial cell function in vitro Studies on viability, proliferation and inflammation, J. of Material Science: Mat. in Medicine 15 (4), 321-325, 2004
- G. Barbolini, A.M. Cats, Nanopathology. Treaty of Histopathology. Ed. Piccin New Libraria Padua ISBN 88-299-1769-9 2006, Chap.1.5 pages 75-80
- Oberdörster G, Sharp Z, Atudorei V, Elder A, Gelein R, Kreyling W, Cox C., Translocation of inhaled ultrafine particles to the brain. Inhal Toxicol. 2004 Jun;16(6-7):437-45
- Elder A, Gelein R, Silva V, Feikert T, Opanashuk L, Carter J, Potter R, Maynard A, Ito Y, Finkelstein J, Oberdörster G. Translocation of inhaled ultrafine manganese oxide particles to the central nervous system. Environ Health Perspect. 2006 Aug;114(8):1172-8. Erratum in: Environ Health Perspect. 2006 Aug;114(8):1178
- Nemmar A, Hoylaerts MF, Hoet PH, Vermylen J. 2003, Size effect of intratracheally instilled particles on pulmonary inflammation and vascular trombosis. Toxic. Appl. Pharmacol. 186, 38-45